Microdosing tirzepatide refers to using smaller than standard prescribed amounts of tirzepatide, a dual GLP 1 receptor agonist and GIP receptor agonist used for blood sugar control and weight management. Interest in low dose tirzepatide continues rising among individuals seeking appetite suppression, metabolic health improvement, and reduced side effects.
Tirzepatide functions as incretin based therapy. Dual incretin action influences insulin sensitivity improvement, gastric emptying delay, and appetite regulation. Clinical trial dosage follows structured titration schedule, gradually increasing weekly injection strength to balance effectiveness and tolerability.
What Microdosing Means in Practice
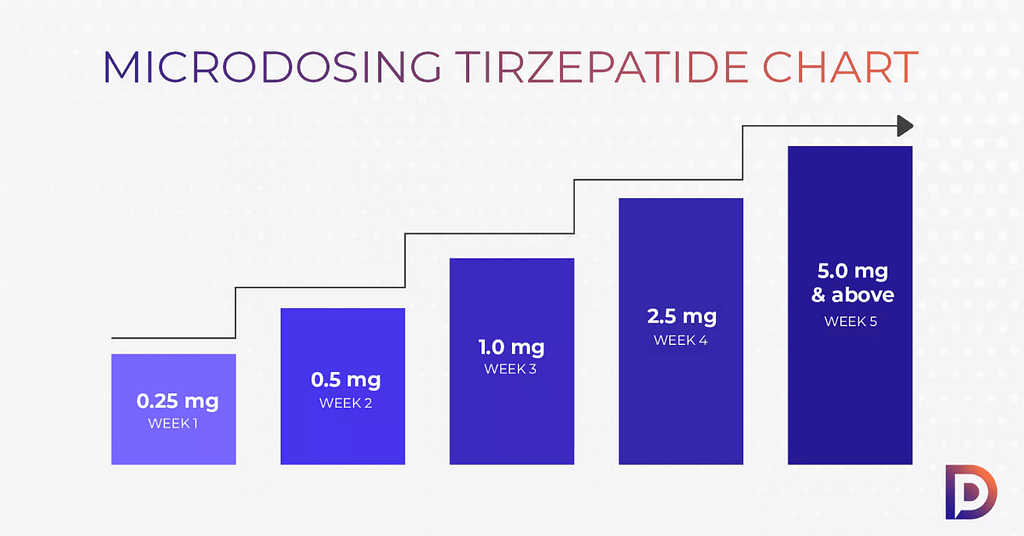
Microdosing tirzepatide typically involves using reduced dose tirzepatide below FDA approved dosage guidelines. Individuals may adjust dosing schedule in attempt to limit gastrointestinal symptoms or improve medication tolerance.
Common motivations include:
• Reduced nausea
• Lower gastrointestinal discomfort
• Gradual metabolic adaptation
• Cost saving strategy
• Appetite control with fewer adverse effects
Medical literature focuses on standard titration schedule tirzepatide. Evidence based dosing emphasizes structured escalation rather than unsupervised small dose tirzepatide use.
How Tirzepatide Works in Body
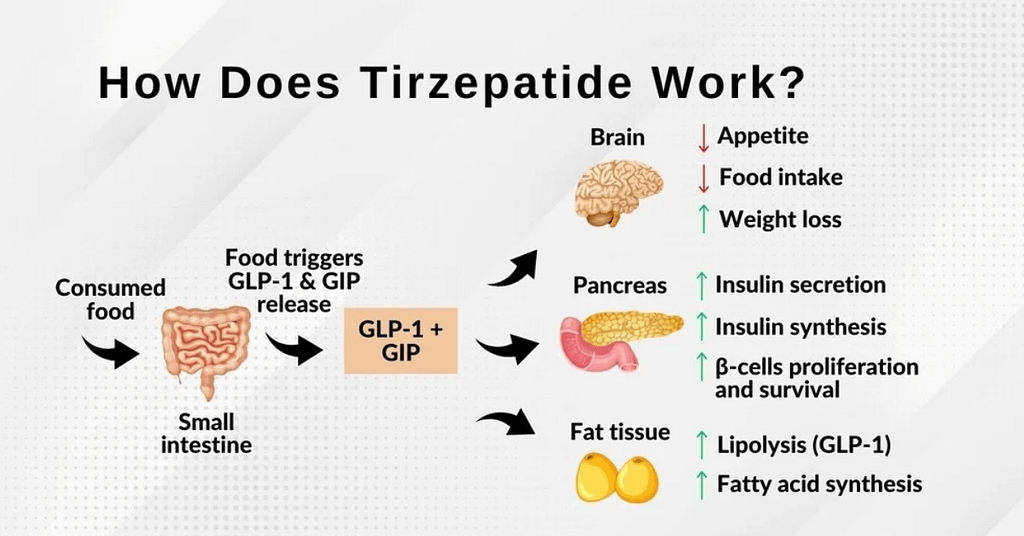
Tirzepatide activates GLP 1 receptor agonist pathway and GIP receptor agonist pathway. This dual incretin therapy enhances insulin secretion, reduces glucagon output, and improves blood sugar regulation medication performance.
Physiological effects include:
• Appetite suppression mechanism
• Improved insulin sensitivity
• Slower gastric emptying
• Enhanced metabolic hormone response
• Fat loss support
These mechanisms contribute toward weight loss injection outcomes observed during clinical trial dosage protocols.
Potential Benefits of Microdosing Tirzepatide
Advocates of microdosing tirzepatide suggest lower dose dependent side effects. Standard dosing sometimes triggers nausea, vomiting, constipation, or injection site reactions. Reduced dose tirzepatide may improve tolerance during early adaptation.
Possible advantages:
• Fewer gastrointestinal symptoms
• Improved medication tolerance
• Gradual appetite suppression
• Lower hypoglycemia risk in some individuals
• Psychological comfort during treatment initiation
However, reduced dose tirzepatide may also limit therapeutic impact. Clinical studies primarily evaluate FDA approved dosage rather than microdose protocol.
Effectiveness for Weight Loss
Tirzepatide for weight loss demonstrates strong results under structured clinical dosing. Obesity treatment medication trials show significant fat loss when following recommended titration schedule.
Microdosing tirzepatide may produce slower or less dramatic weight reduction. Appetite control drug effect depends on receptor activation strength. Subtherapeutic dosing could reduce metabolic impact and limit metabolic health improvement.
Comparison discussions often include semaglutide vs tirzepatide performance. Dual incretin therapy generally provides superior weight loss injection results compared with single GLP 1 medication, yet evidence remains linked to approved dosing.
Safety Considerations
Microdosing tirzepatide involves off label tirzepatide dosing in many cases. Lack of controlled studies evaluating microdose protocol introduces uncertainty.
Safety concerns include:
• Inconsistent blood sugar regulation
• Under treatment of type 2 diabetes
• Pancreatitis warning
• Thyroid tumor warning
• Unpredictable response patterns
Hypoglycemia risk may still occur, especially when combined with other diabetes medication protocol strategies. Endocrinologist recommendation remains essential before any dose adjustment.
Compounded tirzepatide also raises regulatory considerations. Quality control standards vary between compounding pharmacies. Prescription medication oversight ensures appropriate dosing schedule and safety monitoring.
Who Should Exercise Caution
Certain populations require strict medical supervision:
• Type 2 diabetes treatment patients
• Individuals with pancreatitis history
• Family history of thyroid cancer
• Pregnant individuals
• Complex metabolic conditions
Evidence based dosing ensures stable insulin sensitivity improvement and metabolic hormone therapy response. Unsupervised microdosing may compromise treatment consistency.
Cost and Accessibility Factors
Insurance coverage for tirzepatide varies. Brand formulations such as Mounjaro dosage protocols influence financial decisions. Some individuals explore microdosing tirzepatide as cost saving dosing strategy.
Lower weekly injection amounts may extend medication supply. However, long term metabolic outcomes depend on effective receptor activation. Affordable tirzepatide options should be discussed with healthcare provider rather than self directed dose adjustment.
Clinical Perspective
FDA approved dosage recommendations reflect extensive research evaluating efficacy and safety. Clinical trial dosage escalates gradually to reduce gastrointestinal symptoms while maintaining therapeutic strength.
Medication tolerance improves over time during structured titration schedule tirzepatide. Gradual escalation allows incretin hormone response adaptation without compromising weight loss or blood sugar control outcomes.
Microdosing tirzepatide remains under researched compared with established diabetes medication protocol. Healthcare supervision ensures monitoring of metabolic markers, side effects, and treatment goals.
Final Thoughts
Microdosing tirzepatide attracts attention among individuals seeking reduced side effects, flexible dosing schedule, or cost management. Dual incretin therapy offers powerful metabolic benefits, yet strongest evidence supports FDA approved dosage and structured titration schedule.
Low dose tirzepatide may improve tolerability for some individuals, but limited research exists regarding long term effectiveness and safety. Consultation with qualified healthcare professional remains critical before modifying prescription medication strategy.
Safe, evidence based dosing supports sustainable weight management, blood sugar regulation, and overall metabolic health improvement.
For more, you can visit our website
FAQs
Can you microdose tirzepatide?
Microdosing tirzepatide is possible, but it is off label and should only be done under medical supervision.
What should you not do on tirzepatide?
Avoid changing your dose without medical advice and do not combine it with other glucose lowering drugs without guidance.
Is microdosing semaglutide more effective?
There is no strong clinical evidence that microdosing semaglutide is more effective than standard prescribed dosing.
Does tirzepatide lower anxiety?
Tirzepatide is not approved for anxiety treatment, though some users report mood changes during weight loss.